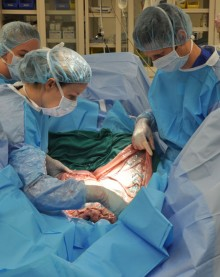
CALEC surgery, or cultivated autologous limbal epithelial cell surgery, marks a groundbreaking advance in treating corneal damage that was previously deemed untreatable. This innovative procedure, pioneered by Ula Jurkunas at Mass Eye and Ear, employs cutting-edge stem cell therapy to repair the cornea’s surface by utilizing the patient’s own ocular stem cells. Over an 18-month clinical trial, patients who underwent CALEC experienced significant restoration of their corneal integrity, offering new hope for those suffering from debilitating blinding injuries. By extracting and cultivating limbal epithelial cells from a healthy eye, the procedure creates a graft that is then transplanted to rejuvenate the damaged cornea. This revolutionary approach not only alleviates visual impairment but also enhances the overall quality of life for many patients affected by corneal damage, harnessing the power of modern science and personalized medicine.
Cultivated Autologous Limbal Epithelial Cells (CALEC) surgery represents a significant leap forward in the realm of ocular medicine, particularly in the realm of corneal restoration. This novel treatment involves harvesting limbal epithelial cells, which are crucial for maintaining the cornea’s smooth surface, and employing them to treat severe corneal injuries. The procedure has garnered attention for its promising results in restoring vision for patients with deep corneal damage. By leveraging advancements in stem cell research and cellular therapies, CALEC surgery stands out as a potential solution for conditions once considered hopeless. Its development by experts at Mass Eye and Ear emphasizes the importance of innovative therapeutic approaches in combating corneal damage and enhancing patient outcomes.
Overview of CALEC Surgery at Mass Eye and Ear
CALEC surgery represents a groundbreaking advancement in the treatment of corneal damage, particularly for patients suffering from limbal stem cell deficiency. Developed and refined at Mass Eye and Ear, this innovative procedure involves extracting limbal epithelial cells from a healthy eye, cultivating them to create a graft, and then transplanting this graft to repair a damaged cornea. The surgery, which was performed for the first time by Ula Jurkunas, has shown a remarkable success rate of over 90% in restoring the corneal surface, offering renewed hope to patients with previously untreatable eye conditions.
The procedure’s significance lies not only in its high success rates but also in its methodology. The use of cultivated autologous limbal epithelial cells allows for a personalized approach to treatment, ensuring that the patient’s own cells are used to promote healing and minimize rejection risks. As this technique pushes the boundaries of traditional corneal repair methods, it serves as a potential model for how stem cell therapy can transform treatments for ocular injuries and other related ophthalmic conditions.
The Role of Stem Cell Therapy in Corneal Repair
Stem cell therapy has emerged as a revolutionary technique in the treatment of various eye-related injuries, especially corneal damage. In the context of CALEC surgery, stem cells play a vital role in regenerating the cornea’s surface, replacing damaged or deficient limbal epithelial cells. The ability to harness the regenerative power of ocular stem cells has opened new avenues for treating conditions once deemed hopeless, such as severe corneal scarring or chronic exposure due to limbal stem cell deficiency.
The results from the clinical trial at Mass Eye and Ear underscore the effectiveness of this treatment, revealing that significant improvements in visual acuity and corneal restoration can be achieved in a patient population that previously lacked viable options. As further studies progress, the hope is that stem cell therapy will not only enhance individual patient outcomes but also pave the way for broader applications across other diseases and injuries affecting the eye.
Mechanism of Cultivated Autologous Limbal Epithelial Cells
The process of cultivating autologous limbal epithelial cells begins with a minimally invasive biopsy from a healthy eye, from which the vital stem cells are extracted. These cells are then expanded in a controlled laboratory environment to form a cellular graft, a procedure that typically takes two to three weeks. This innovative method ensures that the graft is tailored specifically to the patient’s cellular makeup, reducing the likelihood of transplant rejection and fostering a more efficient healing process.
Once the graft is matured, it is surgically implanted into the damaged eye. This meticulous approach not only addresses the immediate corneal surface damage but also aims to restore the natural balance of limbal stem cells, thereby enhancing the eye’s ability to maintain its surface integrity. As researchers at Mass Eye and Ear continue to refine the procedure, they aim to optimize graft production and extend its application to patients suffering from bilateral eye damage, which would greatly increase the pool of beneficiaries.
Impact of CALEC on Vision Restoration
The impacts of CALEC surgery extend beyond mere restoration of corneal integrity; they encompass the broader spectrum of visual rehabilitation. Participants in the clinical trial reported varying degrees of vision improvement, highlighting the potential for significant enhancement in quality of life for individuals plagued by severe visual impairments due to corneal injuries. With restoration rates reportedly reaching 92% at the 18-month follow-up mark, CALEC surgery positions itself as a vital option in ocular regenerative medicine.
As with any emerging treatment, it remains critical to monitor the long-term outcomes of patients undergoing CALEC surgery. Evidence suggests that not only do patients experience improved corneal health, but the overall prognosis regarding visual acuity and patient satisfaction tends to be markedly favorable. Future studies aimed at expanding this treatment’s availability could lead to substantial transformations in how we address and manage corneal diseases.
Challenges and Future Directions of CALEC Surgery
Despite the promising outcomes associated with CALEC surgery, several challenges persist. The requirement for patients to have only one healthy eye to serve as a donor site limits the applicability of this treatment. Furthermore, the manufacturing process of the grafts necessitates specialized facilities and adherence to stringent FDA regulations, which can impede access and scalability. Addressing these challenges is critical to maximizing the benefits of CALEC surgery for a broader demographic of patients.
Looking ahead, researchers are focused on developing an allogeneic manufacturing process that would allow for the use of limbal stem cells from deceased donors. This advancement could significantly democratize access to CALEC surgery, thereby accommodating individuals with bilateral damage or those who lack a suitable donor eye. Collaborations with research institutes and medical facilities nationwide will be essential to navigate these challenges and fulfil the therapeutic potential of cultivated limbal epithelial cell treatments.
Safety Profile of CALEC Surgery
The safety profile of CALEC surgery has been a primary focus of researchers involved in the clinical trials, particularly given the innovative nature of stem cell therapy in ophthalmic applications. Preliminary findings indicate that CALEC surgery is well-tolerated among participants, with a high safety margin observed throughout the various clinical stages. Reports of severe adverse effects were minimal, and any complications that arose appear manageable, suggesting a favorable risk-benefit ratio.
Such robust safety data reinforces the importance of continued research and natural expansion of this treatment option. As additional cohorts are included in future studies, researchers aim to verify these safety results across diverse patient populations, which will help elucidate whether the initial findings hold true in varied clinical scenarios. Ensuring a solid safety profile will be essential as CALEC scopes toward broader clinical application and eventual FDA approval.
Collaboration Between Researchers and Institutions
The remarkable success of CALEC surgery at Mass Eye and Ear is emblematic of the power of collaborative research efforts. Teams comprised of clinicians, scientists, and regulatory professionals worked together seamlessly to bring this innovative treatment from the research bench to the clinic. This multidisciplinary collaboration not only expedited the clinical trial process but also helped to refine protocols for graft production and patient care.
As this research continues to develop, sustaining such partnerships will be pivotal in tackling the numerous challenges associated with advancing stem cell therapies in ocular medicine. Engagement with institutions like Mass Eye and Ear, Dana-Farber, and Boston Children’s Hospital showcases the importance of a united front in overcoming barriers to treatment accessibility and ensuring a streamlined approach to patient recruitment and protocol compliance.
The Role of FDA in Advancing CALEC Surgery
Securing FDA approval is a critical step for CALEC surgery to reach its full potential as a viable treatment option for patients suffering from severe corneal damage. The clinical trial conducted at Mass Eye and Ear represents the first human study of a stem cell therapy targeting the eye in the United States. The meticulous collection of data and patient outcomes during the trial will be essential in demonstrating the therapy’s safety and efficacy to the regulatory body.
As researchers like Jurkunas advocate for continued exploration of CALEC, clear documentation and thorough reporting of patient results will be integral for shaping future discussions with the FDA. Hurdling these regulatory challenges is necessary to bring hope and restoration to patients eager for new solutions to their corneal issues. Through diligent research and advocacy, there is optimism that CALEC surgery can transition from an experimental approach to a mainstream treatment in ophthalmology.
Patient Perspectives on CALEC Treatment
Understanding patient experiences and perspectives regarding CALEC surgery is crucial for assessing the overall impact of the treatment. Early reports from participants in the clinical trial indicate not just improvements in corneal health, but also in overall well-being and quality of life. Living with the complications of corneal damage can be tremendously burdensome, so the ability to regain sight through a procedure like CALEC instills hope and excitement among patients.
Future studies must consider patient-reported outcomes alongside clinical metrics to provide a comprehensive understanding of the treatment’s implications. Engaging with participants through interviews and surveys can yield valuable insights into the emotional and psychological benefits accompanying visual restoration efforts. As CALEC surgery advances, patient feedback will remain a vital element in the ongoing development of stem cell therapies for ocular health.
Frequently Asked Questions
What is CALEC surgery and how does it work?
CALEC surgery, or cultivated autologous limbal epithelial cells surgery, is a groundbreaking treatment developed at Mass Eye and Ear that addresses corneal damage caused by limbal stem cell deficiency. The procedure involves extracting limbal epithelial cells from a healthy eye through a biopsy, cultivating them into a cellular graft in a process that takes two to three weeks, and then transplanting this graft into the damaged eye, effectively restoring the cornea’s surface.
How effective is CALEC surgery in treating corneal damage?
Clinical trials have shown that CALEC surgery is over 90% effective in restoring the corneal surface in patients with blinding corneal injuries. During follow-ups of 12 to 18 months, complete success rates reached 79% and 77%, respectively, indicating this innovative stem cell therapy has a significant impact on restoring vision.
Who conducted the clinical trials for CALEC surgery?
The clinical trials for CALEC surgery were led by Ula Jurkunas, an associate director of the Cornea Service at Mass Eye and Ear. This research marks the first human study of a stem cell therapy involving the eye in the USA, supported by the National Eye Institute, part of the National Institutes of Health.
What types of eye injuries can CALEC surgery help treat?
CALEC surgery is primarily aimed at treating patients with severe corneal damage resulting from various injuries, including chemical burns, infections, and other traumatic events that lead to limbal stem cell deficiency. These injuries often leave the cornea’s surface permanently damaged, making conventional treatments insufficient.
Is CALEC surgery widely available for patients?
Currently, CALEC surgery remains experimental and is not available at Mass Eye and Ear or other U.S. hospitals. Further studies are required before the treatment can be submitted for FDA approval, but researchers at Mass Eye and Ear are optimistic about the future of this stem cell therapy.
What are limbal epithelial cells, and why are they important for cornea health?
Limbal epithelial cells are specialized stem cells located at the limbus, the border area of the cornea. They are crucial for maintaining the smooth surface of the eye and play a vital role in the regeneration of the cornea. Damage that depletes these cells can lead to chronic corneal problems, making treatments like CALEC surgery essential for recovery.
What safety profiles were observed during the CALEC trials?
The phase of CALEC surgery conducted showed a high safety profile, with no serious adverse events reported in donor or recipient eyes. The only noted complication was a minor bacterial infection in one participant, linked to chronic contact lens use, indicating that overall, the procedure is well-tolerated.
What future research is planned for CALEC surgery?
Future research for CALEC surgery plans to involve larger patient cohorts and multi-center studies with randomized controlled designs. This will help in further validating the treatment’s efficacy and supporting its case for FDA approval, ultimately aiming to make this revolutionary therapy accessible to a broader range of patients.
Can CALEC surgery help patients with damage in both eyes?
Currently, CALEC surgery requires the patient to have only one eye affected since it relies on harvesting stem cells from the healthy eye. However, researchers hope to develop an allogeneic manufacturing process that would utilize limbal stem cells from cadaveric donor eyes, potentially expanding the treatment options for patients with bilateral eye damage.
Who are the key researchers involved in the development of CALEC surgery?
The key researchers involved in the development and clinical trials of CALEC surgery include Ula Jurkunas, the principal investigator at Mass Eye and Ear, and Reza Dana, the director of the Cornea Service. Their collaborative work, along with contributions from researchers at Dana-Farber Cancer Institute and Boston Children’s Hospital, has been instrumental in advancing this innovative stem cell therapy.
| Key Points | Details |
|---|---|
| First CALEC Surgery | Performed by Ula Jurkunas at Mass Eye and Ear. |
| Purpose of CALEC Surgery | To repair eye damage, specifically blinding corneal injuries, using stem cell therapy. |
| Clinical Trial Results | Showed 90% effectiveness at restoring corneal surfaces in patients after 18 months. |
| Process Overview | Involves extracting stem cells from a healthy eye, expanding them into a graft, and transplanting into a damaged eye. |
| Safety Profile | High safety with no serious adverse events; minor issues resolved quickly. |
| Current Status | Experimental and not yet available at hospitals, requiring further studies for federal approval. |
Summary
CALEC surgery represents a groundbreaking advancement in ophthalmology, offering new hope for patients with previously untreatable corneal damage. The innovative stem cell therapy has successfully demonstrated its effectiveness in clinical trials, providing a viable solution for restoring the cornea’s surface and significantly improving patients’ quality of life. As research continues, the ultimate goal remains to make CALEC surgery accessible to a broader audience, ensuring that those with severe eye injuries have the opportunity for recovery and improved vision.